Shenzhen Cell Valley and Peking University Deep Research Institute published a high-level article on bispecial CAR T
Recently, Dr. Zhao Lijun, head of the R&D department of Shenzhen Cell Valley, with the first author and Professor Cao Yu of Peking University Shenzhen Graduate School as the corresponding author, published a paper in the internationally renowned journal Protein & Cell (SCI Region 1 journal). Impact Factor 13.6) published A paper "A novel loop-structure-based bispecific CAR that targets CD19 and CD22 with enhanced therapeutic efficacy. against B-cell malignances." This work is an excellent example of the transformation of basic research into industry and clinical application in cell therapy.
Chimeric antigen receptor T (CAR-T) cell therapy has achieved remarkable results in the treatment of B-cell malignancies, but some patients still face the problem of recurrence. In order to solve the heterogeneity of antigen escape and antigen expression, researchers have designed various forms of CAR constructs that recognize double or multiple antigens to reduce the recurrence rate. However, nearly all CD19/CD22 bisspecific Cars reported in recent years have insufficient CD22 targeting, which is caused by the spatial configuration of CD19 and CD22 antibodies.
Most of the reported CD19/CD22 CAR T cells maintain a strong response to CD19 stimulation, but exhibit lower potency under CD22-mediated activity. Considering the potential tonic signal caused by scFv aggregation, we propose a novel CD19/CD22 bisspecific structure (CD19/CD22 LoopCAR-1) that replaces CD22 scFv with CD22 nanoantibodies. CD19/CD22 LoopCAR-1 exhibits better CD22 redirection activity than LoopCAR-4 or other loop-structured CD19/CD22 cars that have been clinically demonstrated, suggesting that LoopCAR-1 may provide geometrically optimal immune synapses (IS). It showed effective double specificity and prominent anti-leukemia activity. This novel structural design provides a unique idea for ring structure design using nanoantibodies and scFv, and may extend the scope of CAR-T cell therapy to solid tumors, as well as indications beyond cancer.

First, the researchers built three different forms of CD19/CD22 cars using G4S Linker, Long Hinge Lama Linker, and β-Stranded Loop Linker, respectively. They are named TanCAR-1, TanCAR-2, and LoopCAR-1. It has been proved that LoopCAR-1 not only significantly enhanced the killing activity of CAR-T on B-ALL patients cells, but also significantly increased the level of cytokine release and promoted cell proliferation. These results suggest that the ring structure may be the best design for bispecific CD19/CD22 cars.
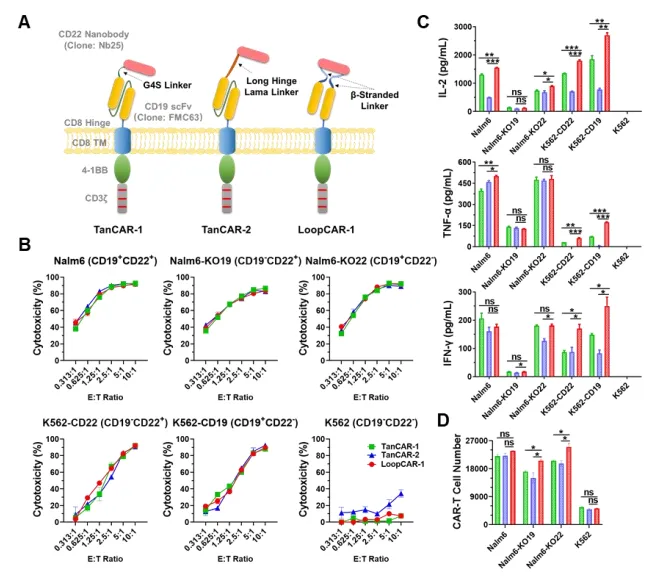
Based on this ring structure, the researchers further designed the CD19/CD22 ring CAR structure by replacing Linker and CD22 nanoantibodies.
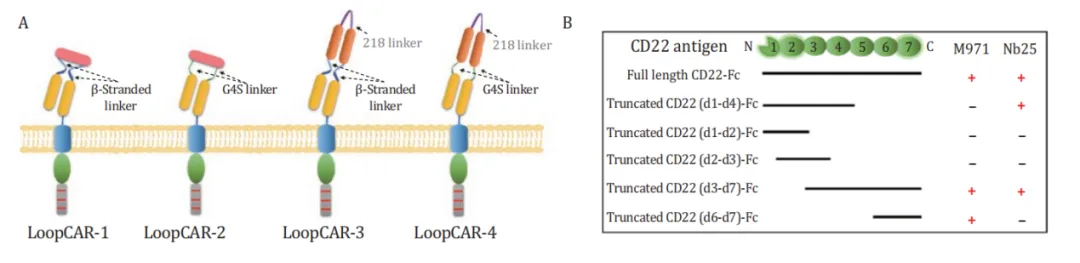
Among the four structures, LoopCAR-1 and LoopCAR-2 had particularly significant effects on Nalm6 and Nalm6 KO19 cells. After two rounds of target cell stimulation, all CAR-T structures showed considerable cytotoxicity, while LoopCAR-1 had more durable cytokilling activity than other bisspecific CARs. In addition, we observed an increase in effector T cell populations after LoopCAR-1 restimulation, but a decrease in the expression of depletion markers, suggesting that LoopCAR-1 has sustained antitumor activity against CD19 antigen-lost target cells. These results show that the selection of antibodies and Linker plays a key role in the design of CAR structure, and LoopCAR-1 is the best CD19/CD22 bispecific CAR structure.

To uate its anti-tumor effect in vivo, the researchers tested the efficacy of each CAR T cell by constructing a xenograft mouse leukemia model using Nalm6 cells (CD19+CD22+). The results showed that the mice treated with LoopCAR-1 did not lose weight and survived significantly longer. In addition, these mice had higher serum cytokine levels than those receiving CD19 CAR, CD22 CAR, or single-target CAR-T combination therapy.These results suggest that in the body of CD19 + CD22 LoopCAR - 1 + tumors have powerful killing effect.
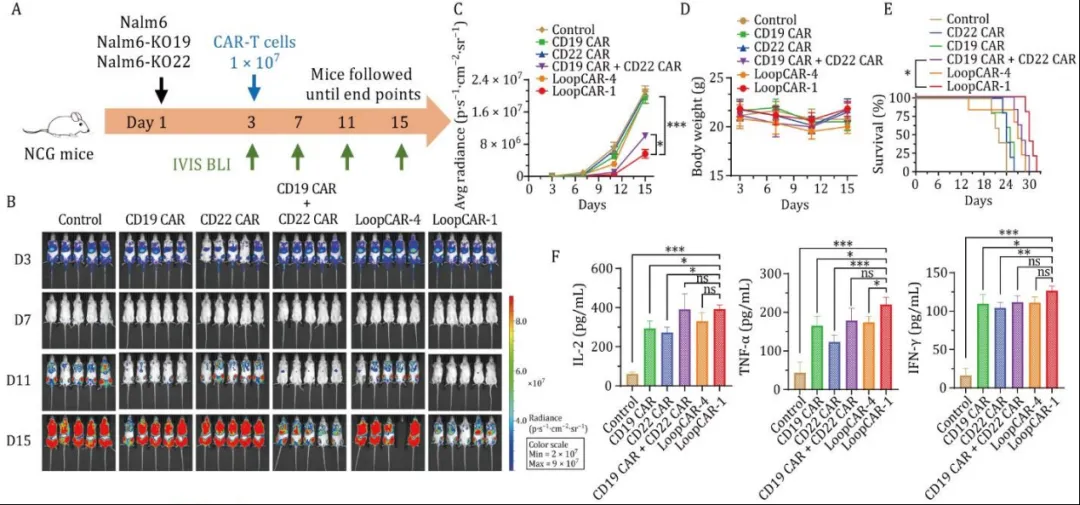
Then, the researchers further uated the performance of LoopCAR-1 in immune escape models with CD19 or CD22 knockout (1:1 ratio) and pseudo-clinical relapse models (1:1:1 ratio), and the results showed that LoopCAR-1 significantly inhibited tumor growth, delayed disease progression, prolonged survival, and increased serum cytokine levels. And did not cause weight loss. However, compared to LoopCAR-1, the combination treatment group with LoopCAR-4 and single-target CAR-T cells performed poorly in reducing tumor burden. This highlights the advantages of the LoopCAR-1 structure. In summary, dual-target CAR-T with LoopCAR-1 structure has the best efficacy against leukemia, preventing immune escape and tumor recurrence associated with mono-specific therapy.

Finally, the researchers uated the efficacy of CAR T cell therapy in a clinically relevant B-ALL PDX model. The results showed that although the mice treated with either ring CAR or single-target CAR combination could not completely eradicate primary B-ALL cells, the LoopCAR-1 treatment group still had stronger anti-leukemia activity, higher overall survival, and more Th1 cytokine production at a later stage compared with other CAR-T cell therapies. In addition, the leukemia cells maintained the expression of CD19 and CD22 after relapse, excluding CAR stress-mediated antigen loss. These results show that LoopCAR-1 has a stable therapeutic effect on the B-ALL PDX model, revealing that it is a promising candidate for clinical treatment.
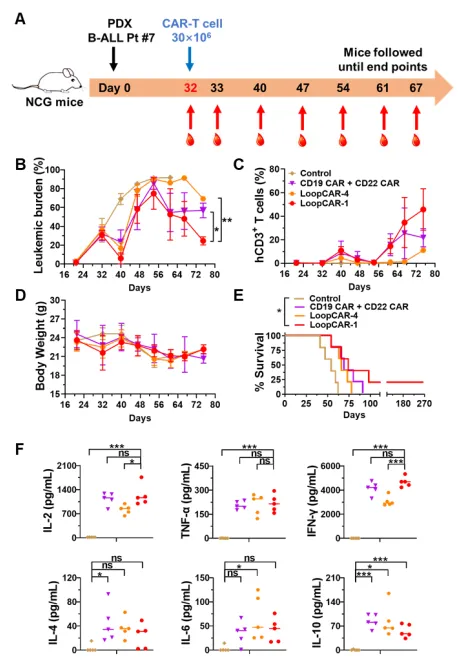
In summary, we developed a novel bispecial CD19/CD22 LoopCAR-T structure that shows significant advantages in enhancing CD22 redirection signaling compared to other cyclic CD19/CD22 cars. The importance of bisecific CAR-T cell structure design and optimization was emphasized by comparing with the clinical CAR-T cells or single-target CAR-T cell combination therapy group. In patients with B-ALL or lymphoma expressing CD19 and/or CD22, LoopCAR-1 has shown more durable clinical value. At present, the novel bifspecificity CD19/CD22 LoopCAR-T has been prepared by Shenzhen Cell Valley and applied in clinic, and has been clinically treated in 4 patients with B-cell malignant tumor in Union Shenzhen Hospital of Huazhong University of Science and Technology (Nanshan Hospital), and the effect is remarkable. Interested readers welcome to contact our company for in-depth exchanges!



